Optimizing Neuropsychiatric Outcomes with Active Folate Supplementation
The Emerging Role of Nutritional Psychiatry
Mental health disorders, particularly anxiety and depression, have a growing global health burden and significant clinical, societal, and economic effects. These disorders are associated with substantial functional impairment, reduced quality of life, and increased healthcare burden worldwide. Although important progress has been made within health care, chronic treatment resistance, poor treatment response, or disease control have emerged, with multiple drug responses, and we should respond in a complementary manner from the biological point of view. Nutritional psychiatry is an emerging field of research with significant scientific relevance to nutrition, metabolism, and brain function. There is now an increasing role for different metabolic pathways, such as folate-dependent one-carbon metabolism, which is vital to neurotransmitter synthesis, methylation processes, and overall neurochemical homeostasis. Disruption of these pathways could lead to poor brain function, depression, and anxiety, and so metabolic optimization is a new and helpful treatment for anxiety and depression.
The Neurobiology of Anxiety and Depression: Beyond Neurotransmitters
Anxiety and depression are increasingly perceived as complex, multifactorial diseases that result in a full complexity not only from altered neurotransmitters but also from a dynamic interplay of neurochemical, metabolic, and molecular mechanisms. Dysregulation of major neurotransmitter species such as serotonin, dopamine, and norepinephrine, which are key to pathophysiology, has recently been shown to involve other causal mechanisms:
At the molecular level, optimal neurotransmitter production and neuronal function are strongly dependent on the availability of methyl donors (primarily from folate-dependent one-carbon metabolism). This pathway is key to the balance of neurotransmitters, synaptic activity, and neuroplasticity. Disruptions in functional methylation and folate metabolism may thus affect neurotransmitter production and brain function, making these pathways mainstays of mental health modulation and targets of therapeutic intervention. In this context, supplementation with biologically active folate (5-methyltetrahydrofolate, 5-MTHF) represents a mechanism-based approach to support methylation processes and maximize neurotransmitter synthesis directly, thus enhancing the clinical outcomes in anxiety and depression.
Folate-Dependent One-Carbon Metabolism in Brain Function
Folate is pivotal to one-carbon metabolism, an integrated network of biochemicals that plays a significant role in maintaining neurochemical equilibrium in the brain, cellular balance, and homeostasis at optimal levels. This pathway is necessary for several vital processes, including:
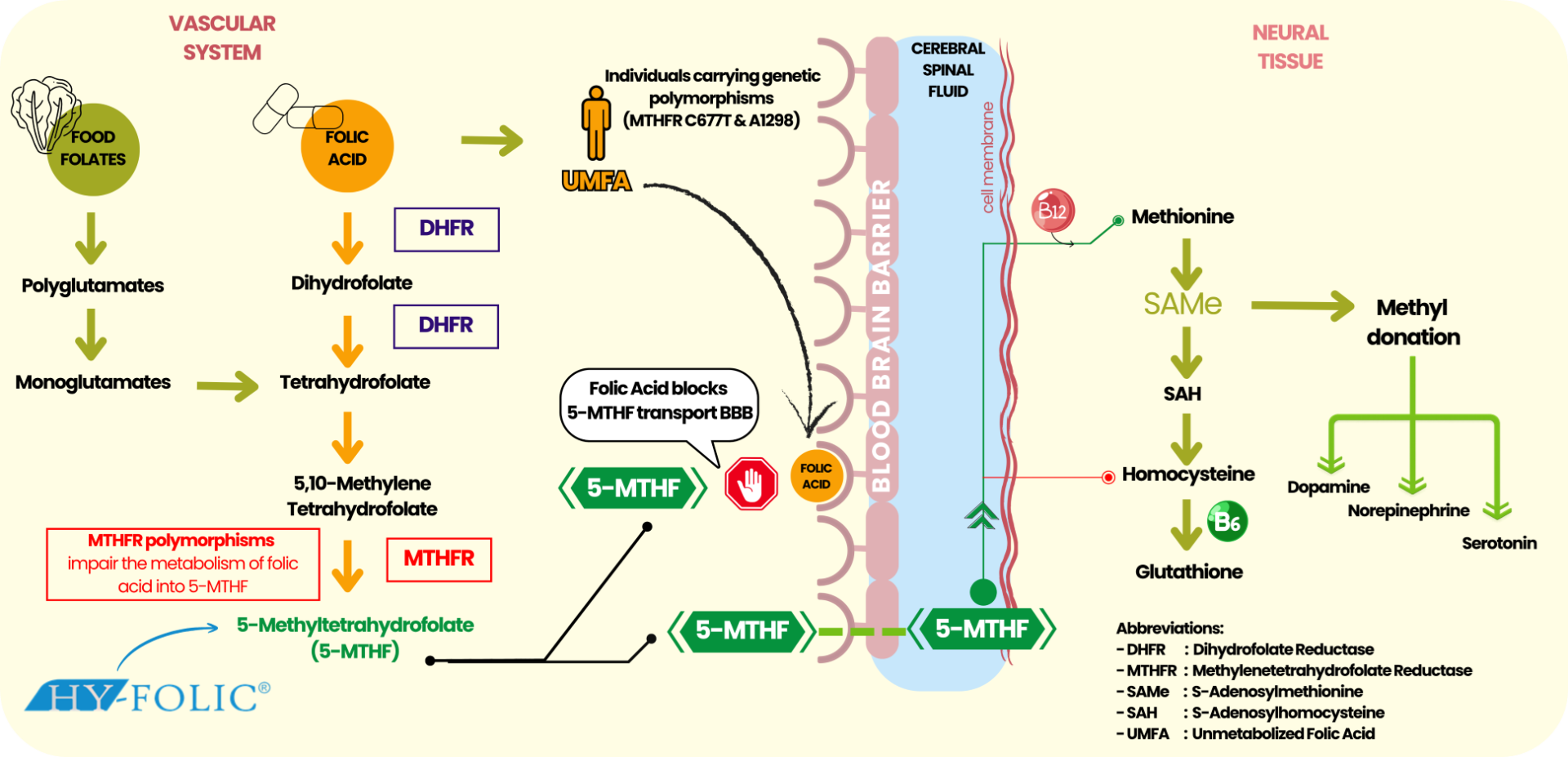
Figure 1. Folate-Dependent One-Carbon Metabolism in Brain Function
One of its major biochemical reactions is homocysteine remethylation to methionine, which subsequently yields S-adenosylmethionine (SAMe), the primary methyl donor necessary for a variety of neurotransmitter, phospholipid, and neuronal protein methylation reactions. Therefore, adequate SAMe supply is crucial for the proper functioning of our brains and healthy neurotransmission. Dysfunction of folate-dependent one-carbon metabolism may have neurobiological effects, such as:
These findings suggest folate-dependent one-carbon metabolism to be a key metabolic regulator of brain function and suggest that this may also serve as a targeted treatment for anxiety and depressive disorders as a key therapeutic intervention target.
Hyperhomocysteinemia and Mental Health Disorders
High levels of homocysteine (hyperhomocysteinemia) have also recently emerged as a metabolic abnormality that is of clinical relevance in relation to anxiety, depression, and cognitive impairment. In addition to acting as a biomarker, homocysteine functionally contributes to chronic neuropsychiatric diseases through a series of different interconnected processes. Molecularly, hyperhomocysteinemia correlates with:
These alterations compromise neurochemical stability, neuroplasticity, and cognitive-emotional regulation, which are fundamental components of the etiology and maintenance of anxiety and depressive illness. Clinical and observational studies show consistent and dramatic decreases in folate content and elevation in plasma homocysteine status in individuals with depression, thus establishing this as a metabolic biomarker of disease burden and a candidate target of therapeutic intervention. In this regard, homocysteine remethylation-mediated interventions, including biologically active folate (5-methyltetrahydrofolate, 5-MTHF), offer a mechanistic approach to restore metabolic homeostasis, support neurotransmitter synthesis, and ameliorate symptoms of anxiety and depression.
Limitations of Dietary Folate and Conventional Folic Acid Supplementation
Folate is a naturally occurring nutrient, mainly in leafy green vegetables and whole grains; however, dietary intake alone is not enough for the greater physiological demand of pregnancy planning. In addition, synthetic folic acid is biologically inactive and must be metabolically activated before it can participate in folate-dependent pathways. This gradual step, however, requires enzymatic completion by dihydrofolate reductase (DHFR) and methylenetetrahydrofolate reductase (MTHFR) to produce the active form, 5-methyltetrahydrofolate (5-MTHF), by biological channels. Despite this, such conversion is intrinsically inefficient and highly heterogeneous at the individual level, especially for those exposed to common MTHFR polymorphisms (C677T and A1298C), which are widely prevalent. Affecting about 25% of the world’s population and 42% of people in Southeast Asia, these genetic variations have a major impact on those with reduced capacity to utilize folic acid effectively. Given that, traditional folic acid supplementation may produce fluctuating clinical response, described as:
These limitations highlight a critical gap in conventional folic acid supplementation, particularly in mental health, where efficient folate metabolism is essential for maintaining neurochemical balance and optimal brain function. Conversely, biologically active folate (5-methyltetrahydrofolate, 5-MTHF) overcomes these limitations by bypassing enzymatic conversion, assuring immediate bioavailability and consistent cellular metabolic activity. Active folate establishes itself as a more predictable and efficient standard in folate supplementation by effectively restoring methylation capacity and finely controlling homocysteine balance, for example, a more efficient approach that could be applied clinically.
Active Folate (5-MTHF): A Mechanism-Based Neuropsychiatric Solution
5-Methyltetrahydrofolate (5-MTHF) is the biologically active form of folate and the predominant circulating form capable of directly participating in central nervous system metabolism. 5-MTHF, in contrast to synthetic folic acid, does not need to be enzymatically activated and can be quickly involved in folate-dependent one-carbon pathways for neurotransmitter homeostasis and optimal neurological functioning. This has implications for several clinical purposes:
Due to these pathways, active folate binds to important metabolic pathways in a major way for neurotransmitter imbalance, impaired methylation, and high homocysteine concentrations associated with anxiety and depression. These features contribute to 5-MTHF development as a precision-controlled mechanism in neuropsychiatric care that also offers a specific neurochemical modality to improve neurochemical balance and clinical outcomes in patients with anxiety and depression.
Clinical Evidence: Active Folate in Anxiety and Depression
Increasing clinical evidence now supports the use of active folate (5-methyltetrahydrofolate, 5-MTHF), a biologically active form of folate, as an effective adjunctive treatment for anxiety and depressive disorders. Active folate directly participates in one-carbon metabolism and promotes methylation, aiding effective neurotransmitter synthesis, neurochemical balance, and overall brain function. Clinical-based studies have also proven that supplementation with 5-MTHF may:
Low folate status, with impaired endogenous methylation capacity. Patients with raised homocysteine levels, consistent with impaired one-carbon metabolism. MTHFR polymorphisms are due to a decreased conversion of folic acid to functional folate. These results support the use of active folate (5-MTHF) as a clinically applicable precision-based adjunct that supports the therapeutic adaptation and the resolution of metabolic substrates for anxiety and depression.
Clinical Applications and Target Populations
Folate supplementation (5-methyltetrahydrofolate, 5-MTHF). Folate is a clinically relevant and mechanism-based approach to neuropsychiatric diseases, especially for patients with metabolic or genetic challenges. Active folate directly improves methylation capacity, neurotransmitter synthesis, and homocysteine metabolism in one-carbon metabolism, which is thus important for treating major biological determinants of mental disorders.
Active folate supplementation would be especially useful for certain populations:
Active folate is a precision-oriented intervention that complements conventional treatment in these populations, enhancing therapeutic response and clinical outcomes.
The Future of Folate Supplementation in Neuropsychiatric
The paradigm for managing mental health is swiftly developing towards precision medicine, blending traditional pharmacotherapy with focused metabolic and nutritional interventions. And that paradigm shift reflects the growing awareness that neuropsychiatric diseases are not only mediated by neurotransmitter dysfunction but also by metabolic derangements that affect brain chemistry, methylation pathways, and neuronal activity. Under this paradigm, biologically active folate (5-methyltetrahydrofolate, 5-MTHF) has an important role here by:
Active folate here is a tailored, mechanism-driven strategy to target specific brain processes to maximize neurochemical homeostasis and impact clinical outcomes among mental disorders (anxiety and depressive disorders). It is thus well-studied based on these mechanisms. Now, in this evolving landscape, PT Simex Pharmaceutical Indonesia offers HY-FOLIC®, a formulation of active folate (5-MTHF) that focuses on a clinically suitable remedy to support neuropsychiatric health and enhance treatment outcomes.
References :