

Folate (vitamin B9) is an essential water-soluble B-vitamin that plays a critical role in human physiology. It is involved in DNA synthesis, nucleotide biosynthesis, cellular proliferation, and epigenetic methylation. Folate also serves as a key cofactor in one-carbon metabolism, a biochemical pathway responsible for maintaining normal cellular function and metabolic homeostasis.
The first generation of folate refers to naturally occurring folate compounds present in food sources, including green leafy vegetables, broccoli, legumes, papaya, mushrooms, eggs, and meat. These forms typically exist as reduced polyglutamate derivatives. However, natural folates are chemically unstable and susceptible to oxidative degradation during food storage and thermal processing. The bioavailability of food-derived folate is estimated to range between 25–50%, depending on the food matrix and preparation method. Experimental studies have shown that folate concentrations in food may decline by up to 70% after three days of storage at room temperature, while cooking processes may lead to losses of up to 95% of the original folate content.
The second-generation of folate is folic acid, a synthetic oxidized form of folate widely used in dietary supplements and food fortification programs. Despite its chemical stability, folic acid is biologically inactive and requires metabolic activation within the body before it can participate in folate-dependent metabolic pathways. This conversion occurs through sequential enzymatic reactions mediated primarily by dihydrofolate reductase (DHFR) and methylenetetrahydrofolate reductase (MTHFR), ultimately producing the active form 5-methyltetrahydrofolate (5-MTHF).
Long-term consumption of high doses of folic acid may lead to the accumulation of unmetabolized folic acid (UMFA) in systemic circulation due to limited enzymatic conversion capacity. Elevated UMFA concentrations have been associated with several potential physiological effects, including:
The third-generation of folate consists of 5-methyltetrahydrofolate calcium salt, which represents the biologically active circulating form of folate. Unlike folic acid, this form does not require metabolic conversion by MTHFR before entering the folate cycle. However, the stability of 5-MTHF calcium salt is temperature sensitive, with optimal stability observed under refrigerated conditions (2–8°C). This limitation may reduce its practicality in tropical climates where maintaining cold-chain storage is challenging.
The fourth-generation of folate represents an advancement in folate formulation technology. It consists of 6S-5-methyltetrahydrofolate glucosamine salt (5-MTHF glucosamine salt), commonly referred to as active folate. The incorporation of glucosamine as a counterion improves the chemical stability, solubility, and bioavailability of the active folate molecule compared with earlier folate formulations.
Key characteristics of 5-MTHF glucosamine salt (Active Folate) include:
A supplement containing 5-MTHF glucosamine salt 1,100 mcg is available in the product HY-FOLIC®.
Folic acid and 5-methyltetrahydrofolate (5-MTHF) represent chemically and metabolically distinct forms of vitamin B9. Folic acid is a synthetic inactive precursor commonly used in supplements and fortified foods. It requires multiple metabolic steps before conversion into its active metabolite, 5-MTHF. By contrast, 5-MTHF represents the predominant physiological form of folate circulating in human plasma and can be directly utilized in cellular methylation reactions. However, metabolic conversion of folic acid to 5-MTHF does not occur with equal efficiency in all individuals. Incomplete metabolism may lead to the accumulation of unmetabolized folic acid (UMFA) in the bloodstream.
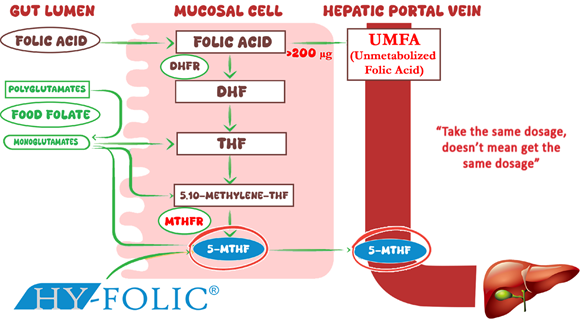
Figure 1. Differential Metabolism of Folic Acid and 5-MTHF in the Human Folate Cycle
Variations in the MTHFR (methylenetetrahydrofolate reductase) gene significantly influence folate metabolism. Common polymorphisms such as C677T and A1298C can reduce MTHFR enzymatic activity and impair the conversion of folic acid into its active form. Epidemiological studies indicate that:
carry MTHFR polymorphisms that may affect folate metabolism. Data from the 1000 Genomes Project further demonstrate that genetic variants affecting folate metabolism are relatively common worldwide. These findings suggest that a uniform folic acid supplementation strategy may not be optimal for all individuals, particularly those carrying MTHFR variants. Providing folate directly in its active form (5-MTHF) may offer a more efficient metabolic pathway for improving folate status and reducing plasma homocysteine concentrations.
HY-FOLIC® provides folate directly as 5-MTHF, thereby bypassing the MTHFR-dependent conversion step and allowing immediate metabolic utilization.
A pharmacokinetic study conducted through collaboration between PT Simex Pharmaceutical Indonesia and the Institute of Medical Research and Education (IMERI), Faculty of Medicine, Universitas Indonesia, evaluated the pharmacokinetic profile of HY-FOLIC® compared with folic acid in healthy Indonesian volunteers. The study demonstrated that plasma concentrations of active folate following HY-FOLIC® administration were approximately two-fold higher than those observed after folic acid supplementation, indicating superior systemic bioavailability.
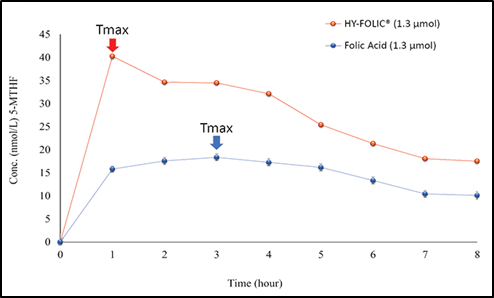
Figure 2. Comparative Pharmacokinetic Study of HY-FOLIC® and Folic Acid in Healthy Volunteers.
5-MTHF represents the predominant physiological form of folate in human circulation. Approximately 91% of total circulating folate in plasma exists as 5-MTHF. Similarly, 5-MTHF constitutes the majority of folate detected in maternal serum and neonatal cord blood, representing approximately:
These findings highlight the importance of adequate folate availability during pregnancy, as folate plays a fundamental role in fetal growth, neural development, and the prevention of neural tube defects. Adequate folate intake from early life stages is also important for reducing the risk of impaired growth and developmental outcomes, including early childhood stunting.
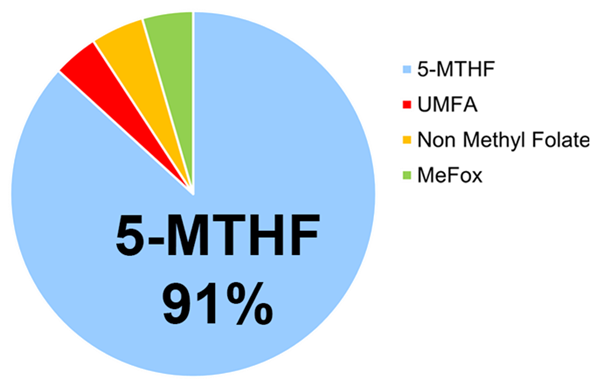
Figure 3. Relative Distribution of Folate Forms in Human Blood
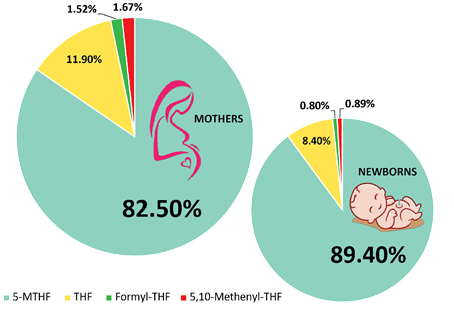
Figure 4. Relative Distribution of Folate Forms in Maternal Serum and Neonatal Cord Serum
Active folate supplementation may be beneficial for individuals with increased physiological folate requirements or impaired folate metabolism, including:
1. Couples planning conception, to support optimal preconception folate status.
2. Pregnant and lactating women, due to increased maternal and fetal folate demands.
3. Patients with cardiovascular disorders, particularly those associated with elevated homocysteine levels.
4. Individuals with renal impairment, in whom folate metabolism may be altered.
5. Patients with neurodegenerative disorder (Alzheimer's, Parkinson's, cognitive impairment), and other neurological disorders (epilepsy, peripheral neuropathy).
6. Individuals with psychiatric conditions, including mood disorders (depression and bipolar), psychotic disorders (schizophrenia), anxiety disorders, and neurodevelopmental disorders (ASD, ADHD).
Recent advances in the understanding of folate metabolism, genetic polymorphisms, and one-carbon methylation pathways have revealed important limitations associated with conventional folic acid supplementation. Increasing scientific evidence suggests that metabolic variability, particularly related to MTHFR genetic polymorphisms, may influence the efficiency of folic acid conversion into its biologically active form.
In this context, supplementation with 5-methyltetrahydrofolate (5-MTHF) represents a more physiologically relevant strategy for optimizing folate status. As the predominant circulating form of folate in human plasma, 5-MTHF can be directly utilized in methylation reactions and homocysteine metabolism without requiring enzymatic activation.
HY-FOLIC® provides folate in the form of 5-MTHF, a stabilized formulation of active folate designed to enhance bioavailability, metabolic efficiency, and clinical applicability. By delivering folate in its biologically active form, HY-FOLIC® supports immediate metabolic utilization and may represent an advanced approach for improving folate status across diverse populations, including individuals with impaired folate metabolism.
HY-FOLIC® contains 5-MTHF, the biologically active form of folate, which does not require multiple metabolic conversion steps
Peak plasma concentrations of HY-FOLIC® are approximately two-fold higher than those observed with folic acid supplementation
Because 5-MTHF does not require enzymatic activation, supplementation does not lead to accumulation of unmetabolized folic acid (UMFA)
As the understanding of folate metabolism, genetic variability, and one-carbon metabolism continues to advance, biologically active forms of folate such as 5-methyltetrahydrofolate (5-MTHF) have emerged as a physiologically relevant strategy for optimizing folate status and supporting metabolic and developmental health. In this context, HY-FOLIC® from PT Simex Pharmaceutical Indonesia, which provides folate directly in its active form, may represent a scientifically supported option for addressing these metabolic challenges.