Transforming Vascular Cognitive Health Through Active Folate Supplementation
The Growing Burden of Vascular Cognitive Impairment
Vascular cognitive impairment (VCI) is a vast family of cognitive disorders that can have a variety of clinical features and range from subtle cognitive impairment to severe vascular dementia due to cerebrovascular pathology. It accounts for a significant and growing burden of global cognitive morbidity, especially among ageing populations where the magnitude of vascular risk factors continues to increase. VCI also often occurs alongside other neurodegenerative diseases, compounding disease burden and speeding up functional decline. At the clinical level, VCI is characterised by ongoing impairment in executive function, attention, and memory, secondary to cerebral hypoperfusion, microvascular injury, and neurovascular impairment.
These mechanisms suggest that vascular dysfunction and neuronal damage interact in a multifactorial manner, placing VCI as a multifactorial disorder at the intersection of vascular and neural pathology. Despite progress in treating cardiovascular risk factors, treatments are mostly symptomatic and do not address the underlying pathophysiological mechanisms. This clinical gap highlights the need for mechanism-based, disease-modifying therapeutics that target major mechanisms of neurovascular dysfunction, including core metabolic pathways such as homocysteine regulation and folate-dependent one-carbon metabolism, to more effectively prevent and slow cognitive decline.
The Neurovascular Metabolic Link in Cognitive Decline
Part of the cognitive decline in vascular cognitive impairment (VCI) involves the interrelated neurovascular metabolic pathway of vascular dysfunction, metabolic imbalance, and neuronal injury. And without it, brain perfusion, cellular health, and ultimately brain functioning would be affected. At the vascular level, endothelial dysfunction is paramount, impairing vasodilation, reducing nitric oxide bioavailability, and compromising cerebral blood flow regulation. As a consequence, chronic cerebral hypoperfusion hampers oxygen and nutrient delivery to neural tissue, which leads to neuronal stress and degeneration. At the same time, neuroinflammation and oxidative stress serve as major pathological amplifiers.
Excess generation of reactive oxygen species (ROS) and inflammatory mediators induces vascular injury, blood-brain barrier disruption, and neuronal toxicity, which, in turn, hastens the progression of cognitive decline. These vascular and inflammatory processes are also mediated by metabolic dysregulation, with folate-dependent one-carbon metabolism and homocysteine accumulation implicated. It establishes a bidirectional cycle of damage, with vascular damage worsening neuronal impairment and neuronal damage promoting vascular disruption. Taken together, the dynamic interplay between vascular impairment and neuronal failure suggests targeting the neurovascular metabolic axis as a strategically effective means to preserve cognitive function and slow disease progression.
Folate-Dependent One-Carbon Metabolism in Neurovascular Health
Folate plays a master role as an important factor in the one-carbon metabolic network, a highly organized set of interrelated biochemical pathways necessary for neurovascular health, cell homeostasis, and brain health. This pathway controls the transfer of one-carbon units, which is necessary for many biological processes that link vascular function with brain nerve function. Folate-dependent one-carbon metabolism in the neurovascular system is highly integrated with:
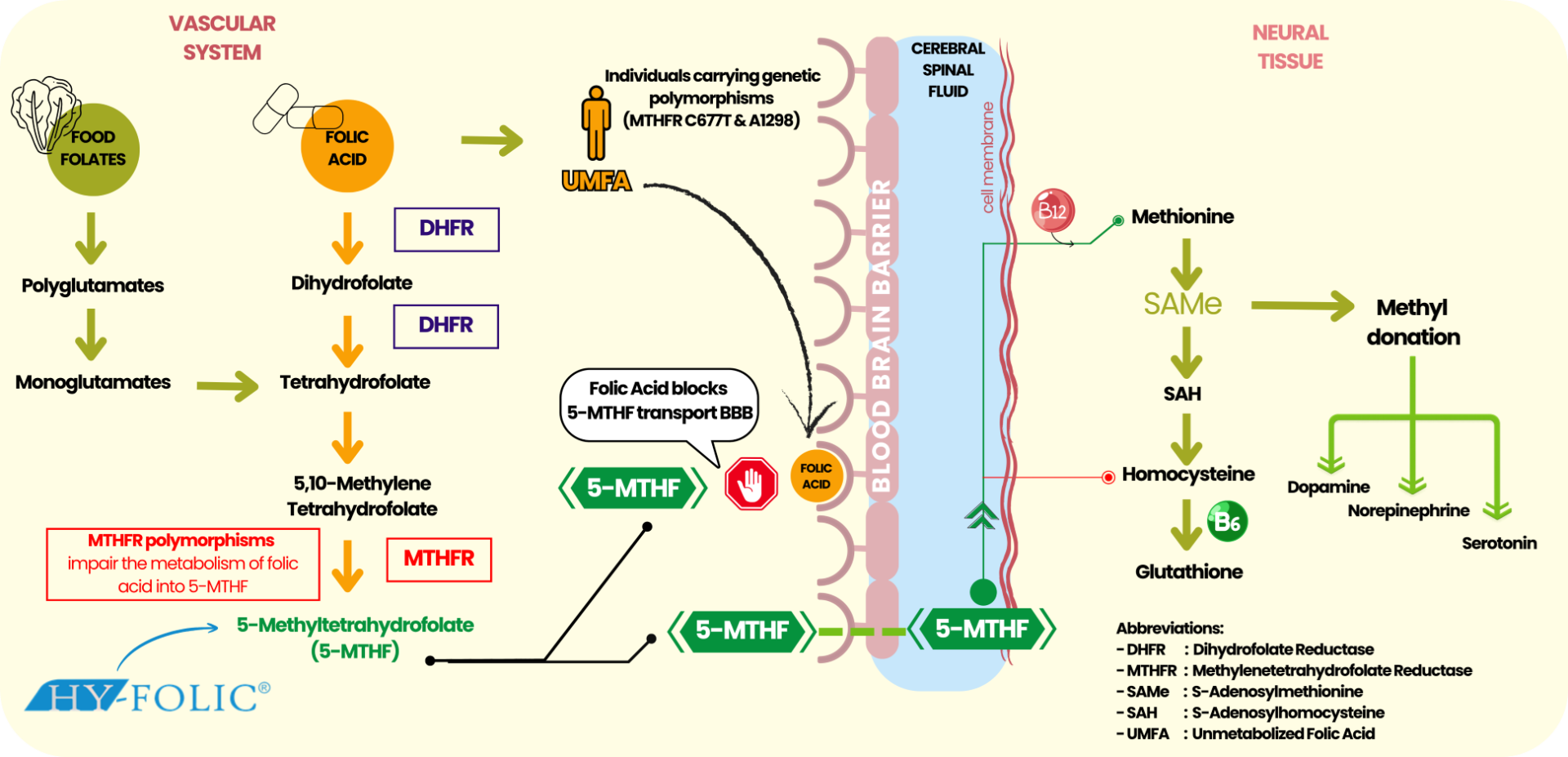
Figure 1. Folate-Dependent One-Carbon Metabolism in Neurovascular Health
One major biotechnological step in this pathway is the remethylation of homocysteine to methionine, which yields S-adenosylmethionine (SAMe), the primary methyl donor for DNA, RNA, protein, and lipid methylation. Maintaining endothelial function, vascular tone, and efficiency of neuronal communication is critically important to adequate SAMe availability. These pathways form a central regulatory architecture that maintains interconnectivity between vascular health and cognitive function within the one-carbon pathway. Disruption of this pathway may affect methylation capacity, cell repair, and neurotransmission, leading to neurovascular dysfunction. Perhaps the most prominent clinical effect of impaired folate metabolism is the accumulation of homocysteine, a metabolite that is detrimental to vascular and neuronal tissues; hyperhomocysteinemia thus plays a major role in driving cognitive decline.
Hyperhomocysteinemia as a Modifiable Risk Factor in Cognitive Decline
Hyperhomocysteinemia, an abnormally high circulating homocysteine, is both a clinically important and modifiable metabolic risk in the pathogenesis of vascular cognitive impairment (VCI) and dementia. As a mediator in folate-dependent one-carbon metabolism, homocysteine acts as the focal link between metabolic derangement and vascular and neurological dysfunction. Both at the molecular and vascular levels, hyperhomocysteine is a pathogenic contributor to neurovascular injury through numerous pathways:
These effects compromise cerebral perfusion and vascular homeostasis and decrease neuronal viability, thereby hastening cognitive decline. Clinically, there is overwhelming evidence that high concentrations of homocysteine are linked with an increased risk of cognitive impairment, stroke, and dementia, with an association between higher levels of homocysteine and increased rates of cognitive decline. Hyperhomocysteinemia is not a non-modifiable risk factor, as it represents a modifiable metabolic pathway that may be important for targeted therapeutic interventions. Optimization of homocysteine metabolism through folate-based pathways, especially biologically active folate (5-methyltetrahydrofolate, 5-MTHF), offers a mechanism-based approach to restore vascular function while preserving neuronal integrity and, therefore, aiding cognitive preservation.
Limitations of Dietary Folate and Conventional Folic Acid Supplementation
Folate is a naturally occurring nutrient, mainly in leafy green vegetables and whole grains; however, dietary intake alone is not enough for the greater physiological demand of pregnancy planning. In addition, synthetic folic acid is biologically inactive and must be metabolically activated before it can participate in folate-dependent pathways. This gradual step, however, requires enzymatic completion by dihydrofolate reductase (DHFR) and methylenetetrahydrofolate reductase (MTHFR) to produce the active form, 5-methyltetrahydrofolate (5-MTHF), by biological channels. Despite this, such conversion is intrinsically inefficient and highly heterogeneous at the individual level, especially for those exposed to common MTHFR polymorphisms (C677T and A1298C), which are widely prevalent. Affecting about 25% of the world’s population and 42% of people in Southeast Asia, these genetic variations have a major impact on those with reduced capacity to utilize folic acid effectively. Given that, traditional folic acid supplementation may produce fluctuating clinical response, described as:
These limitations highlight a critical gap in conventional folic acid supplementation, particularly in vascular cognitive health, where efficient folate metabolism is essential for maintaining neurochemical balance and optimal brain function. Conversely, biologically active folate (5-methyltetrahydrofolate, 5-MTHF) overcomes these limitations by bypassing enzymatic conversion, assuring immediate bioavailability and consistent cellular metabolic activity. Active folate establishes itself as a more predictable and efficient standard for folate supplementation by effectively restoring methylation capacity and finely controlling homocysteine balance, thereby offering a clinically applicable approach.
Active Folate (5-MTHF): A Mechanism-Based Approach to Neurovascular and Cognitive Health
5-Methyltetrahydrofolate (5-MTHF) is the biologically active folate that can directly modulate vascular and neuronal pathways, contributing to vascular cognitive impairment (VCI). In contrast to conventional folic acid, 5-MTHF is immediately biologically available, directly involved in one-carbon metabolism, and can help correct metabolic dysfunction. This brings us some clinically significant advantages on the neurovascular side:
Such diverse mechanisms enable active folate to deliver a neurovascular and metabolic boost by targeting metabolic processes in vascular dysfunction and in cognitive decline. The 5-MTHF strategy is a highly specific, mechanism-driven approach to vascular and brain health. It is a solution for cognitive function, helping preserve cognitive status in persons with vascular cognitive impairment (VCIs).
Clinical Evidence and Cognitive Outcomes
A growing stream of clinical and epidemiological research has identified an essential role for folate status and homocysteine regulation in cognition and cognition-related cognitive dysfunction, especially in the setting of vascular cognitive impairment (VCI) and age-related cognitive decline. Efforts to directly target folate-dependent one-carbon metabolism, particularly when they occur with biologically active folate (5-methyltetrahydrofolate, 5-MTHF), have been shown to produce dramatic reductions in plasma homocysteine levels, an important modifiable risk factor for vascular dysfunction and cognitive impairment. In addition to biochemical benefits, clinical literature indicates that improving folate status might lead to quantifiable effects in cognition, in relation to:
These effects are thought to be mediated by improved endothelial function, increased cerebral perfusion, reduced oxidative damage, and restoration of methylation capacity, all of which are necessary for maintaining neuronal structure and cognitive health. The clinical value of active folate is most acute among high-risk groups, such as:
These results underscore active folate as a clinically significant mechanism and treatment-based intervention that not only leads to enhanced metabolic biomarkers but also to enhanced functional cognition, and point to its role in holistic neurovascular rehabilitation of health.
Clinical Applications and Target Populations
Biologically active folate (5-methyltetrahydrofolate, 5-MTHF) supplementation provides a clinically pertinent, mechanism-driven therapy to people at higher risk of neurovascular dysfunction and cognitive decline, especially those with underlying metabolic and vascular vulnerabilities. Active folate could be very beneficial for the following patients:
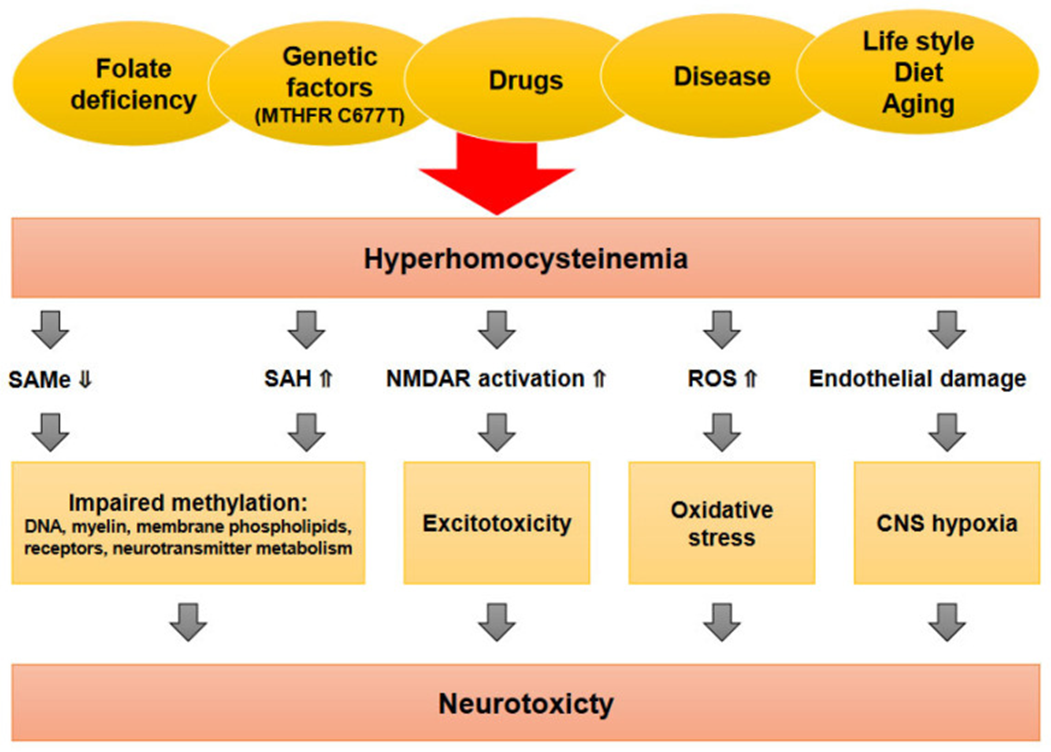
Figure 2. Elevated Homocysteine Levels are Associated
In such populations, active folate provides a targeted metabolic intervention that can interfere with pivotal underlying pathways, thereby promoting vascular integrity and cognitive function.
The Future of Folate Supplementation in Vascular Cognitive Health
The future of cognitive and vascular health is quickly advancing towards precision medicine through the integration of conventional therapeutic strategies with targeted metabolic and nutritional modalities. This represents a paradigm shift, emphasizing that cognitive decline prevention and its treatment must be guided by metabolic dysregulation and cannot be confined to structural and hemodynamic mechanisms, and offering a fundamental insight into how metabolic dysfunctions underpin neurovascular integrity and cerebral function. In this nascent paradigm, biologically active folate (5-methyltetrahydrofolate, 5-MTHF) comes into focus and is a key factor that facilitates:
Active folate thus combines multiple domains of regulation, in line with precision medicine, and serves as a multifaceted mechanism for addressing key biological contributors to neurovascular dysfunction. As neurovascular and neuropsychiatric care continue to move towards personalization and a metabolic-based approach, active folate (5-MTHF) has arisen as an essential element of precision neurovascular nutrition, leading clinical practice with HY-FOLIC® derived from PT Simex Pharmaceutical Indonesia, providing a focused solution to enhance neurochemical balance, vascular health, and cognitive outcomes.
References :